In the field of environmental engineering, treating high-concentration ammonia nitrogen wastewater remains a significant challenge. Traditional biological treatment methods often struggle when faced with complex and diversified water quality. Consequently, immobilized microbial technology has gained widespread application due to its ability to increase relative microbial concentrations and enhance biological treatment efficiency.
As the most commonly used embedding agent for this technology, Polyvinyl Alcohol (PVA) stands out for its low cost, high mechanical strength, and resistance to microbial decomposition. However, native PVA has exposed several "pain points" in practical applications, such as biological toxicity to microorganisms, low recovery rates, and high water-solubility expansion (swelling). To address these issues, researchers are exploring surface crosslinking modification to comprehensively optimize PVA performance.
1. Why Modify PVA?
While native PVA has good film-forming and fiber-forming properties, its stability in water is relatively poor, often leading to swelling that can destroy the integrity of the immobilized membrane. By introducing a crosslinking agent, a reaction is triggered between the agent and the abundant hydroxyl groups in the PVA molecules, constructing a stable network.
PVA has a wide variety of crosslinking agents, such as maleic acid, formaldehyde, and glutaraldehyde (GA). Among these, GA has become a mainstream choice because it operates under mild conditions and does not require heat treatment to drive the reaction. Furthermore, the introduction of Graphene Oxide (GO) is a stroke of genius. GO possesses a massive specific surface area and rich oxygen-containing functional groups, which significantly improve the mechanical properties and chemical stability of the composite material.
2. Experimental Breakdown: From Graphene Oxide to Magnetic Gel Beads
This research utilized a rigorous process to create a high-strength, easily recoverable material:
- Polyvinyl Alcohol 1788 (PVA 1788) Selection: The study utilized PVA 1788 (degree of polymerization: 1788; molecular weight: 84,000–89,000 g/mol; minimum alcoholysis: 87.4%) as the base polymer.
- Preparation of Graphene Oxide (GO): Using an improved Hummers method, natural graphite was oxidized in three stages (low, medium, and high temperature) using concentrated sulfuric acid and potassium permanganate. This expands the graphite layers to create functionalized GO.
- Glutaraldehyde (GA) Modification: To reduce swelling, a 5% PVA solution was reacted with GA to trigger an acetalization reaction.
- Magnetization (MGO-PVA): To solve recovery issues, Fe3O4 magnetic nanoparticles were incorporated into the GO matrix via co-precipitation. This allows the material to be easily recovered using an external magnetic field.
- Gel Bead Preparation: The modified PVA-GA solution was mixed with 1% sodium alginate and specific microbial strains (e.g., ammonia-oxidizing bacteria), then crosslinked in a saturated boric acid and calcium chloride solution.
3. Results and Data Analysis
Through SEM, XRD, and various physical performance tests, the study reached the following core conclusions:
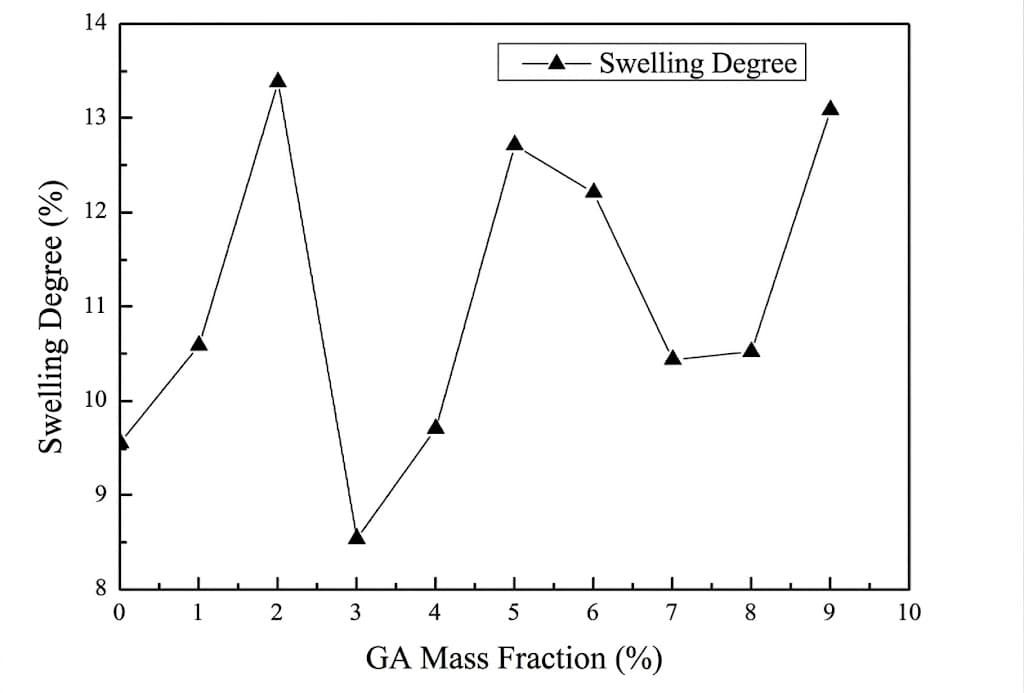
Optimization of Swelling: The 3% Critical Point
The experiment found that when the mass fraction of GA was 3%, the water content of the modified PVA reached its lowest point (8.524%), and the swelling degree was significantly reduced. This indicates that GA successfully reacted with the PVA, reducing the number of hydrophilic hydroxyl radicals and enhancing the material's stability in water.
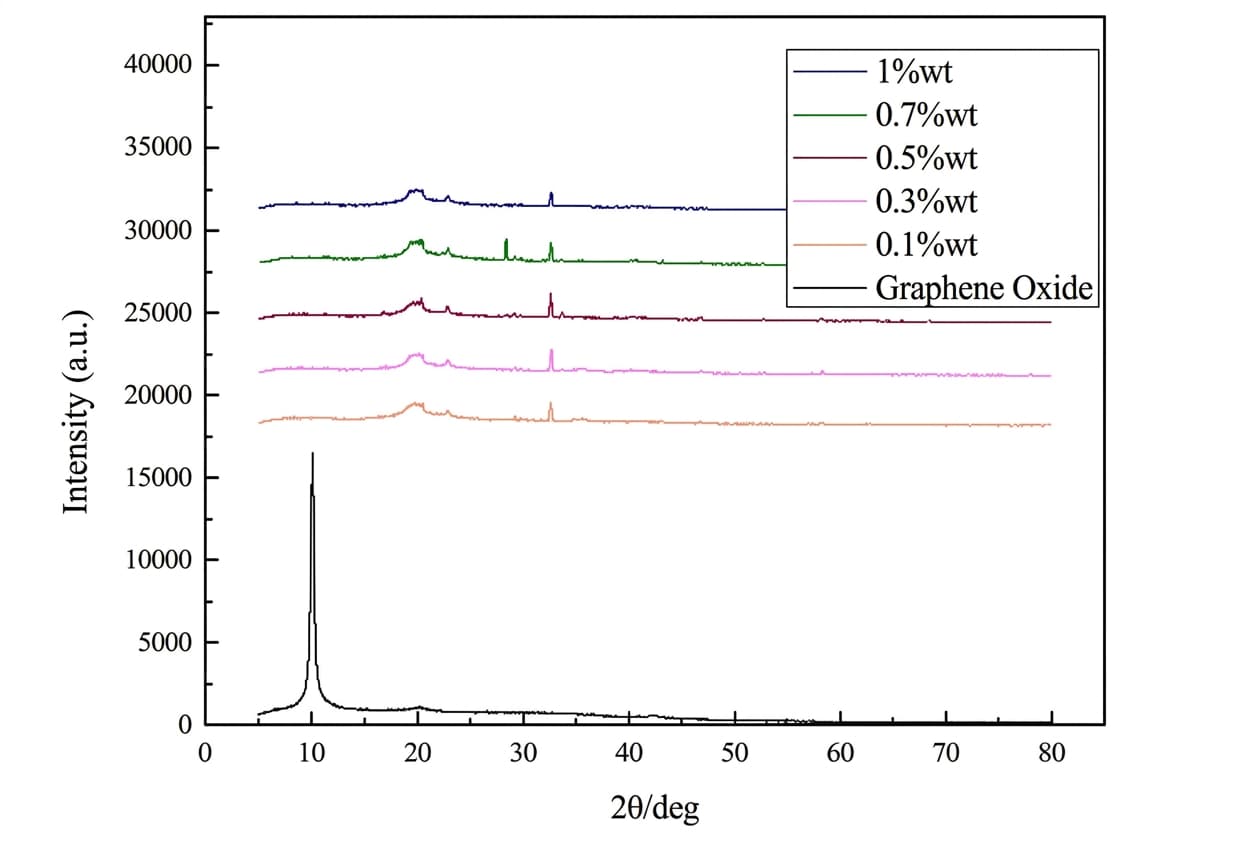
Structural Verification: Successful Magnetization
XRD characterization showed a sharp FexO diffraction peak at approximately 2θ = 32.61°, confirming high crystallinity of the synthesized magnetite. As GO content increased, the typical GO peak at 2θ = 10.09° weakened, proving that GO was uniformly dispersed and successfully integrated with the PVA.
Mechanical Strength and Bounce Performance
In high-speed oscillation tests at 200 r/min, gel beads with 0.3 wt% GO addition performed the best:
- Fragmentation rate was 0%.
- Average bounce range reached 18–23 cm.
This suggests that the 0.3 wt% ratio allows the gel beads to offset hydraulic shear and compression forces through their own elasticity while maintaining sufficient hardness for resistance.
4. Mass Transfer Performance: Ensuring Microbial Respiration
For immobilized microorganisms, mass transfer performance determines whether nutrients can smoothly enter the interior of the beads. Tests showed that beads with 0.1 wt% and 0.3 wt% GO achieved the fastest wetting speed (100%). This indicates that low concentrations of GO help form developed pores, thereby ensuring high mass transfer efficiency.
This research not only provides a new pathway for Modified Polyvinyl Alcohol (Modified PVA) but also directly serves the critical environmental need for high-concentration ammonia nitrogen wastewater treatment.
Website: www.elephchem.com
whatsapp: (+)86 13851435272
E-mail: admin@elephchem.com